What Is Bimagrumab? The Experimental Weight-Loss Drug Scientists and Doctors Are Talking About
Bimagrumab, an experimental obesity drug from Eli Lilly, shows promise in burning fat while preserving muscle. Here’s how it works, trials, results, and timeline.
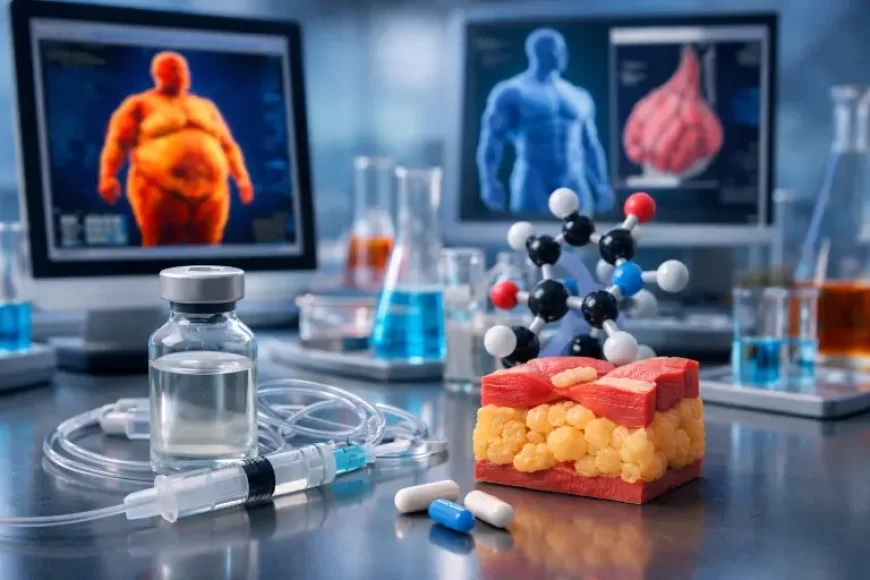
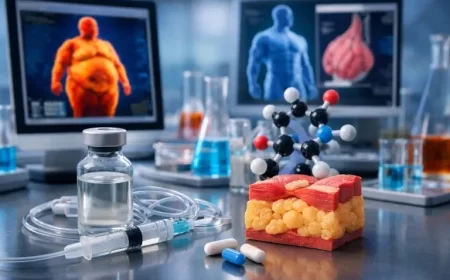
The global race to develop safer and more effective obesity treatments is accelerating. One experimental therapy gaining attention is Bimagrumab, a drug currently in clinical trials that aims to reduce body fat while maintaining — and even increasing — muscle mass.
Unlike popular weight-loss injections that mainly suppress appetite, Bimagrumab works through a different biological pathway. Early studies suggest it could change how obesity and metabolic diseases are treated in the future.
What Is Bimagrumab?
Bimagrumab is a laboratory-engineered monoclonal antibody designed to improve body composition. The drug was originally developed by Novartis and later acquired by Eli Lilly through its purchase of Versanis Bio.
Researchers are exploring the treatment primarily for:
- Obesity
- Metabolic disorders
- Muscle preservation during weight loss
Type 2 diabetes–related body composition issues
The biggest promise of the drug is that it may help people lose fat without sacrificing muscle, a challenge that most weight-loss treatments struggle with.
How Bimagrumab Works in the Body
Blocking Muscle Growth Inhibitors
Bimagrumab targets proteins known as Activin Type II receptors (ActRIIA and ActRIIB). These receptors normally respond to signals like myostatin, which limits muscle growth.
By blocking these signals, the drug allows the body to stay in a muscle-building state, even during fat loss.
Encouraging Fat Burning
Researchers also believe the treatment may influence metabolic pathways that activate fat-burning processes in the body. This helps reduce stored fat while maintaining lean mass.
This dual effect — reducing fat while protecting muscle — is why some scientists refer to it as a potential “holy grail” therapy for body composition.
Clinical Trial Results So Far
Early clinical trials have produced results that caught the attention of obesity researchers.
In a 48-week study involving patients with type 2 diabetes:
Participants lost around 21% of their body fat
Lean muscle mass increased by about 3.6%
Most of the weight reduction came from fat rather than muscle
Typically, traditional weight-loss methods cause both fat and muscle loss. The difference seen with Bimagrumab suggests it could address one of the biggest limitations of current obesity medications.
Combination With Popular Weight-Loss Drugs
Pharmaceutical companies are also exploring how Bimagrumab could complement existing obesity drugs.
Recent research programs are testing the therapy alongside tirzepatide, a widely used GLP-1 based treatment. The goal is to reduce the muscle loss sometimes associated with appetite-suppressing medications.
If successful, the combination approach could significantly improve long-term metabolic health outcomes for patients.
How the Drug Is Given
Unlike many modern weight-loss injections that patients administer at home, Bimagrumab is delivered in a clinical setting.
Administration details:
- Given through intravenous (IV) infusion
- Typically once every four weeks
- Administered under medical supervision
Because of this method, it is currently limited to research settings.
Reported Side Effects
So far, clinical trials suggest the drug is generally manageable, though side effects have been reported.
Commonly observed reactions include:
- Muscle spasms
- Diarrhea
- Acne
Temporary increases in certain enzyme levels
Researchers continue to monitor safety as trials expand.
Current Status of Bimagrumab in 2026
As of early 2026, Bimagrumab is still under investigation and has not been approved for public use.
Key points about its development:
- The drug remains in Phase II/III clinical trials
- Some earlier research programs were discontinued for strategic reasons
- New trials focusing on obesity without diabetes are ongoing
More detailed results are expected in late 2026
Until regulatory approval is granted, the therapy can only be accessed through official clinical studies.
Why Scientists Are Paying Attention
The obesity drug market is currently dominated by GLP-1 medications. While effective for weight loss, these treatments often lead to reductions in muscle mass along with fat.
Bimagrumab approaches the problem from a body composition perspective, which could make it valuable for:
- Long-term metabolic health
- Aging populations
- Patients trying to preserve muscle strength
- Medical weight-loss programs
If future studies confirm the early findings, it could represent a major shift in obesity treatment strategies.
The next two years will likely determine whether Bimagrumab becomes a major medical breakthrough or remains a niche experimental therapy.
With several ongoing trials and interest from a major pharmaceutical company, the drug is expected to generate significant data in the near future. Experts say its real potential will become clearer once larger and longer studies are completed.
For now, it remains one of the most closely watched developments in the evolving field of obesity medicine.
FAQ About Bimagrumab
What is Bimagrumab used for?
Bimagrumab is an experimental drug being studied for obesity, metabolic health, and body-composition improvement.
Is Bimagrumab approved for weight loss?
No. As of 2026, the drug has not been approved by regulators and is only available in clinical trials.
How is Bimagrumab different from Ozempic or Wegovy?
While GLP-1 drugs mainly reduce appetite, Bimagrumab focuses on blocking muscle-growth inhibitors to reduce fat while preserving muscle.
Who is developing Bimagrumab?
The drug was originally created by Novartis and is now being developed by Eli Lilly after acquiring Versanis Bio.
When could Bimagrumab become available?
If ongoing trials are successful, experts expect potential regulatory review later in the decade.
What's Your Reaction?
 Like
0
Like
0
 Dislike
0
Dislike
0
 Love
0
Love
0
 Funny
0
Funny
0
 Angry
0
Angry
0
 Sad
0
Sad
0
 Wow
0
Wow
0